

Structure biology in cellular signaling and membrane trafficking
National Chung Hsing University Assistant Professor 2024.08.01~
Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP) Post-Doctoral Researcher 2018.04-2024.07.31
Genomic Research Center, Academia SINICA Research Assistant 2009.07-2012.04
PI Conversion X Small GTPases:
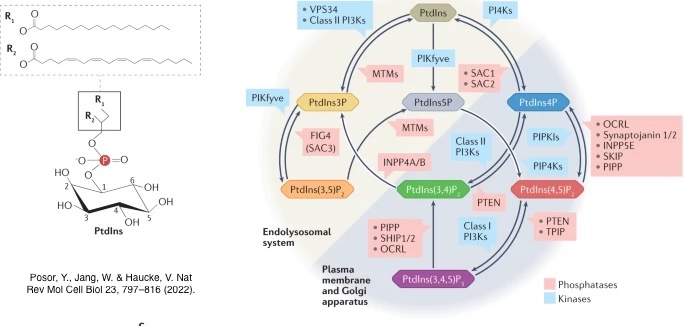
Cells are the basic units of life, with membranes and organelles coordinating communication through lipid signals and their interacting proteins. Among these, phosphoinositides (PIs) are key signaling molecules that regulate many cellular processes.
Seven PI species exist and can be interconverted by specific kinases and phosphatases. Their spatial distribution acts as a “molecular code” that defines organelle identity and, together with over 150 small GTPases, precisely controls cellular signaling.
Disruption of PI regulation is linked to major diseases, including cancer, diabetes, and neurological and muscle disorders. Understanding how these lipid signals are controlled therefore has significant potential for therapeutic development.
Research Interests
Our research focuses on lipid signaling and membrane trafficking, with an emphasis on phosphoinositide regulation and Class II PI3-kinases. These kinases exist as three distinct isoforms, each with unique subcellular localization and functions, and represent promising therapeutic targets for diseases such as cancer, diabetes, thrombosis, stroke, and rheumatoid arthritis.
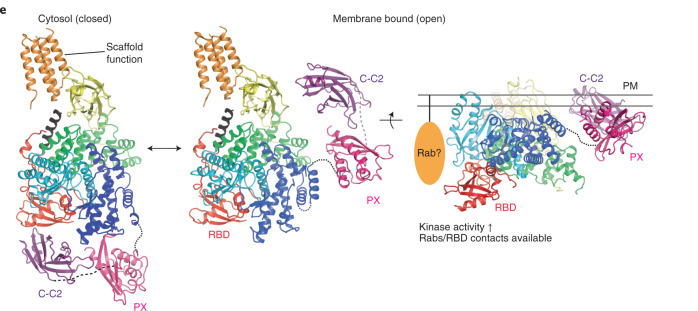
Structural studies of the Class II PI3K α isoform (PI3KC2α) have revealed a general mechanism of autoinhibition shared across Class II PI3Ks. However, key questions remain unresolved, including how these enzymes are activated on membranes and how their lipid signals are terminated.
To address these challenges, our lab investigates phosphoinositide-converting enzymes—including Class II PI3Ks, MTM/MTMR phosphatase complexes, and INPP4A/B—by integrating proteomics, biochemistry, and structural biology. We aim to understand how these enzymes are regulated by small GTPases, such as Rab proteins, to control spatial and temporal signaling dynamics.
Ultimately, our goal is to translate mechanistic insights into therapeutic strategies, including the development of small-molecule inhibitors and nanobody-based approaches to modulate phosphoinositide signaling. These efforts hold broad potential for treating human diseases, including cancer progression and metastasis.
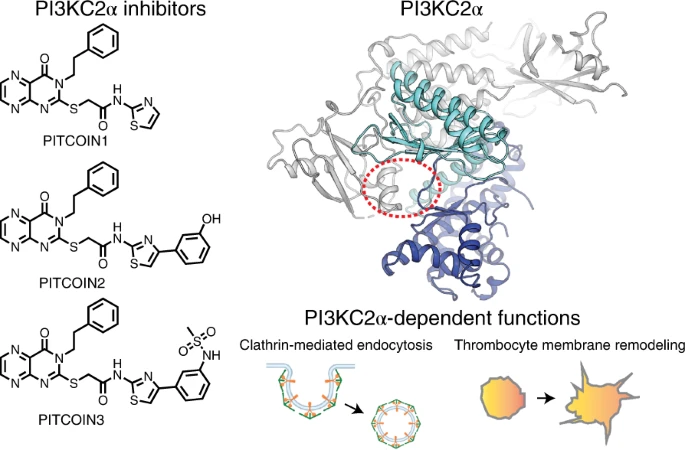
In collaboration with the Leibniz-FMP (Berlin), we develop small-molecule inhibitors guided by protein engineering and X-ray crystallography. This approach has enabled the discovery of PI3KC2α-selective inhibitors (PITCOINs), which are being applied to study endocytosis, cancer cell migration, thrombosis, and cell division. We are further expanding these efforts to develop PI3KC2β inhibitors with potential applications in stroke therapy.
Research Approaches
Our research integrates multi-scale approaches to understand phosphoinositide signaling:
-
Cell biology: intracellular vesicle trafficking and cell division
-
Proteomics: APEX-based proximity labeling using stable cell lines
-
Structural biology: X-ray crystallography, single-particle cryo-EM, and HDX-MS/MS
Freie Universität Berlin (Germany) Chemistry and Biochemistry Dr. rer. Nat (Ph.D) 2012.06-2018.03
National Chung Hsing University Institute of Biochemistry Master of Science 2005.09-2007.07
National Kaohsiung Normal University Department of Chemistry Bachelor 2001.09-2005.06
2024 Yushan Young Scholar
Duhay V, Tian M, Kosieradzka K, Ebner M, Lo WT, Krauss M, Sprengel HL, Voss M, Riechmann M, Savas JN, Schwake M, Haucke V, Damme M. Control of lysosome function by the GTPase-activating protein TBC1D9B and its binding partner TMEM55B. Nat Commun. 2026 Mar 14.
Hoffmann L, Duchmann M, Lazarow K, Huang YH, Lukas F, Lo WT, Feil R, Schmied C, Lehmann M, Lunn J E., Piazza I, Kries J P, Haucke V, Maritzen T. Fructose-1,6-bisphosphate couples glycolytic activity to cell adhesion. Nat Cell Biol 2026 Mar 16.
Lo WT#, Belabed H#, Kücükdisli M, Metag J, Roske Y, Prokofeva P, Ohashi Y, Horatscheck A, Cirillo D, Krauss M, Schmied C, Neuenschwander M, von Kries JP, Médard G, Kuster B, Perisic O, Williams RL, Daumke O, Payrastre B, Severin S, Nazaré M, Haucke V. Development of selective inhibitors of phosphatidylinositol 3-kinase C2α. Nature Chemical biology. 19(1):18-27. 2023.01
Lo WT, Zhang Y, Vadas O, Roske Y, Gulluni F, De Santis MC, Zagar AV, Stephanowitz H, Hirsch E, Liu F, Daumke O, Kudryashev M, Haucke V. Structural basis of phosphatidylinositol 3-kinase C2α function. Nature Structural and Molecular Biology. 29(3):218-228. 2022.03
Samsó P, Koch PA, Posor Y, Lo WT, Belabed H, Nazare M, Laporte J, Haucke V. Antagonistic control of active surface integrins by myotubularin and phosphatidylinositol 3-kinase C2β in a myotubular myopathy model. Proc Natl Acad Sci USA (PNAS) 119(40): e2202236119. 2022.10
Li H, Prever L, Hsu MY, Lo WT, Margaria JP, De Santis MC, Zanini C, Forni M, Novelli F, Pece S, Di Fiore PP, Porporato PE, Martini M, Belabed H, Nazare M, Haucke V, Gulluni F, Hirsch E. Phosphoinositide Conversion Inactivates R-RAS and Drives Metastases in Breast Cancer. Advanced Science. Mar;9(9):e2103249. 2022.03
Gulluni F, Prever L, Li H, Krafcikova P, Corrado I, Lo WT, Margaria JP, Chen A, De Santis MC, Cnudde SJ, Fogerty J, Yuan A, Massarotti A, Sarijalo NT, Vadas O, Williams RL, Thelen M, Powell DR, Schueler M, Wiesener MS, Balla T, Baris HN, Tiosano D, McDermott BM Jr, Perkins BD, Ghigo A, Martini M, Haucke V, Boura E, Merlo GR, Buchner DA, Hirsch E. PI(3,4)P2-mediated cytokinetic abscission prevents early senescence and cataract formation. Science. 374(6573):eabk0410. 2021.10
Wang H#, Lo WT#, Haucke V. Phosphoinositide switches in endocytosis and in the endolysosomal system. Current Opinion in Cell Biology. 59:50-57. 2019.08